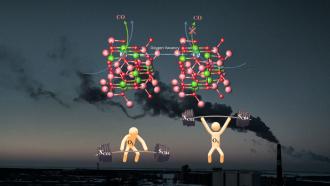
Oxygen is vital for life processes to take place. We wouldn’t last even a few minutes without it. But it so happens that this very oxygen can also cause harm. Oxygen is required mainly so that cellular respiration can occur. This respiration releases much needed energy for running the body. But the by-products of respiration and other oxygen requiring reactions in the body are ‘free radicals’ like superoxide (O2-) and hydrogen peroxide (H2O2). All atoms have a central positively charged core, surrounded by a cloud of negatively charged electrons – something like a miniature solar system. The electrons are usually in pairs orbiting the core. Free radicals are those atoms, molecules or ions which have one or more unpaired electrons in their orbits. These unpaired electrons make these radicals so highly reactive that they can cause damage to cells and tissues. In order to neutralize these harmful free radicals, our bodies produce molecules called ‘anti-oxidants’ which are enzymes. We also get anti-oxidants like vitamins C, A and E, from fruits and vegetables that we eat.
It is thought that free radicals are the cause of aging and can also cause or aggravate diseases like cancer and coronary heart disease. However, this is a highly debated topic and experiments have been conducted to show that this may not be true.