Image credits: ScientiFickly
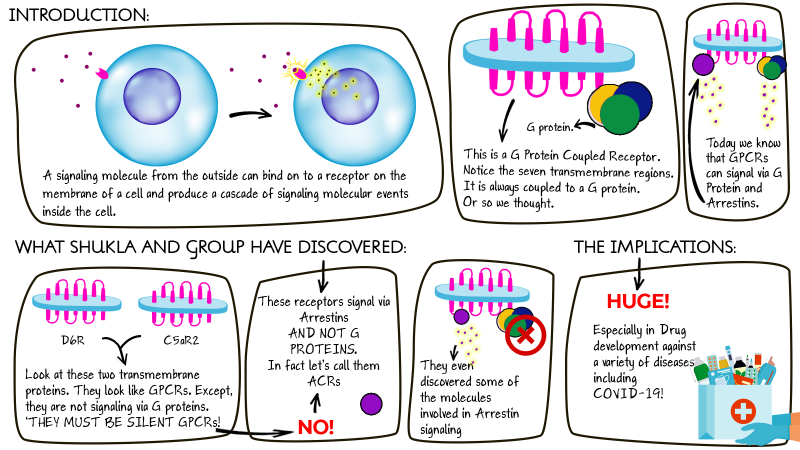
Living cells use various molecular signalling mechanisms to communicate. A plethora of biomolecules, such as proteins, protein fragments and fat molecules, help our body coordinate with different other parts. A receptor is a molecule that sits on a cell membrane and looks out for a signal. A glucose molecule, a hormone, or even a piece of a pathogen can bind to a cellular receptor and initiate signalling from the outside of the cell to the inside. The binding induces a cascade of molecular events that trigger various physiological actions within the cell.
In a new study published in the journal Molecular Cell, a group of researchers have shown that two receptors previously thought to not participate in signalling—hence called ‘silent receptors’—signal via a molecule that was once believed to stop signalling! The discovery can revolutionize the design of drugs against many conditions including inflammation and cancer.
A primary class of receptors to respond to cellular signals are G protein-coupled receptors, called GPCRs. They are involved in almost every bodily process, from seeing and smelling to heartbeats and immunological responses and more. These receptors signal using G-proteins aka guanine nucleotide-binding proteins. All GPCRs are structurally distinctive—they are long-chain proteins with the chain traversing a cell’s membrane inside out exactly seven times. A signal molecule binds to a GPCR on the cell membrane on the outside. The GPCR gets activated and stimulates a specific G protein inside. The G-protein initiates further signalling interactions inside the cell, prompting a particular response from the cell.
In the current research, Arun K Shukla and his group from the Indian Institute of Technology Kanpur, in collaboration with scientists from Tohoku University (Japan), McGill University (Canada) and Queensland University (Australia), have discovered that two receptors that look like conventional GPCRs, hence assumed to be GPCRs, do not signal via the conventional G Proteins. Instead, they use a protein called Arrestin. Textbooks will need to be re-written! Just so you know, Arrestins were initially believed to bind to receptors and arrest signalling; hence the name – Arrestins.
Not all GPCRs work by the textbook understanding of inducing G proteins alone. Some GPCRs convey signals via G proteins and Arrestins, both. And now, with this study, we know that some receptors believed to be GPCRs, work by Arrestin signalling and not G protein-mediated signalling.
The researchers were studying a type of receptors called C5a peptide receptors that include a pair of structurally similar receptors, C5aR1 and C5aR2, which respond to a pathogen attack by causing inflammation to protect the body. During a pathogen attack, our body, in a frenzied state of distress, produces various molecules that induce signalling in different cells, inciting various actions against the intruder. C5a peptide is one such molecule. It binds to both C5aR1 and C5aR2. Scientists have shown that C5aR1 initiates intracellular signalling via G proteins. C5aR2, also a GPCR, on the other hand, did not seem to signal via G proteins. Hence, C5aR2 was considered a silent receptor. But how can C5aR2, structurally similar to C5aR1, not produce the same intracellular signalling response like C5aR1 when activated by the same stimulus? This was the question that led the group down the path to the discovery of Arrestin signalling in C5aR2.
“We knew that blocking these receptors (D6R and C5aR2) produced significant physiological changes. But nobody knew how because these receptors were not signalling via the classical G protein pathway. We wanted to understand the mechanism behind these physiological changes, which is why we dug deeper,” explains Shukla.
Shukla and his team have shown that the two specific GPCRs they studied, D6R and C5aR2, both of which were classified as non-signalling GPCRs, could transmit a signal through Arrestins, but not G Proteins. The current discovery highlights a non-classical signalling mechanism induced by two receptors that are structurally similar to GPCRs.
“When we found the signalling capability of these receptors, it was a kind of ‘Eureka’ moment, especially since it has been over 20 years of recognising them as ‘silent’. No one knew they actually signal via an alternate mechanism” says Shubhi Pandey, a PhD student in Dr Shukla’s laboratory and one of the first authors of this study.
Using various experiments, the team has elucidated how the signalling with Arrestins propagates and some of the molecular events involved. But why is this discovery important?
Almost half the current drugs in the market, including drugs for hypertension, asthma, allergy, acidity, cancer and more, target GPCRs. The drugs work by blocking the GPCR region that can stimulate the G protein – then, the cell will fail to respond to the signal. This is medically useful, especially if the signal triggers the cells to produce, say, an allergic reaction.
“The signalling and the physiological responses mediated by G proteins and Arrestins are very distinct, very different from each other. Blocking a GPCR could block both the signalling pathways, and this lack of specificity gives rise to the side effects of many of the GPCR targeting drugs,” Shukla elaborates. Knowledge of this signalling pathway, as elucidated by Shukla and his team, will help design drug molecules that are specific to either G protein or Arrestin mediated signalling and reduce possible side effects. It could also assist in drug discovery against both pathways at once, if required.
Let’s take the example of C5aR1 and C5aR2 in a coronaviral attack scenario. Following the viral attack, both the receptors are activated and begin signalling using G protein and Arrestin, respectively, resulting in an inflammatory response. Suppose the situation were to worsen and result in rapid and excessive inflammation. In that case, it could also lead to multiple organ failures and eventually death.
Scientists are currently trying to block the interaction of the C5a peptide with C5aR1 as a potential therapy in certain COVID-19 cases. The understanding from the present study could now allow the development of drugs targeting C5aR2, in addition to C5aR1.
“The next move is to understand the structural aspects and how this specific interaction of Arrestins with these two different receptors drives different signalling responses in the cell so that we can identify drug molecules that can bind these receptors,” acknowledges Pandey, referring to future directions.
This article has been run past the researchers, whose work is covered, to ensure accuracy.