Photo : Dr. Rita Khanna
Contemporary history can be characterised by an ever-increasing dependence on modern gadgets to carry out our daily tasks – be it machines that wash our clothes or do the dishes, or a sophisticated personal computer which acts as a window to the world. The growth of distribution networks, regular upgrades and lower prices of these devices have made them more affordable than ever before, and have resulted in a new waste stream: e-waste.
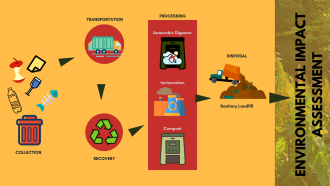
Electrical and electronic devices, discarded at the end of their useful lives, end up as e-waste. Dismantled devices are either recycled, incinerated and/or landfilled. As most of these devices contain some amount of precious metals like gold and silver, several formal and informal e-waste recycling sectors are trying to recover these valuables. However, many of the techniques used are not environmentally sustainable and could release toxic gases during recycling. A recent study by researchers from the Indian Institute of Technology, Roorkee, The University of New South Wales, Sydney and Council of Scientific and Industrial Research (CSIR), Odisha, have proposed a novel approach, to capture toxic emissions generated during the recycling of e-waste, with specific focus on the informal sector.
While the recycling of e-waste has economic benefits, inappropriate treatment processes themselves create a fair share of environmentally harmful outcomes, substantially high in the informal sector. In the Indian context, “the informal sector consists of Kabadi waalas who collect waste from door-to-door, and then sort it in unorganised facilities, without any proper safety procedures and organisation”, points out Prof Rita Khanna, lead researcher in the study.
Various e-waste recycling methods used in the informal sector include manual dismantling of discarded devices, open burning, chipping or melting them, burning wires to recover copper, acid and cyanide salt leaching, and other inadequate metallurgical treatments. These activities release dust particles loaded with potentially toxic elements (PTEs) into the atmosphere that may re-deposit near the emission site, or be transported over long distances depending on the particulate size. Potentially toxic elements (PTEs) constitute elements such as Zinc (Zn), Copper (Cu), Nickel (Ni), Cadmium (Cd), Lead (Pb), Mercury (Hg), Chromium (Cr), Molybdenum (Mo), Selenium (Se) and Arsenic (As).
This study has focussed mainly on capturing such PTEs, emitted during the heat treatment of e-waste. While thermal treatment of e-waste in developed countries uses extensive gas cleaning filters to captures PTEs, these are rarely used in the informal sectors or in developing countries due to inadequate legislation, cost factors and technological challenges. In this study, the researchers used a surface phenomenon – adsorption - to capture PTEs in inorganic adsorbents.
During adsorption, atoms, ions or molecules from a substance (gas, liquid or dissolved solid) adhere or stick to the adsorbent surface, e.g., the use of silica gel in a desiccator to adsorb moisture. The process of adsorption involves two components –the adsorbent and the adsorbate. Adsorbent is the substance on the surface of which adsorption takes place and the adsorbate is the substance which is being adsorbed.
These researchers chose alumina as the adsorbent medium due to its porosity, surface area and capacity for adsorption. Alumina is also hydrophilic (has little affinity for water) and has low tendency to adsorb organic compounds. It is also thermally stable and does not react or decompose at high temperatures around 600⁰C. The researchers tested alumina with particle sizes ranging from 20-45µm to greater than 53µm for their adsorption capacity.
The experiments began by mechanically crushing the printed circuit boards (PBCs) from discarded mobile phones to sizes less than 1mm and then carried out experiments on their thermal degradation at about 600⁰C for different heating times. They observed that metal fumes, dust particles and hazardous constituents could be captured by using adsorbents like alumina. The use of inert gases in the process prevented oxidation during the heat treatment. The adsorption was found to improve with increasing contact time between the gases and the adsorbent. From a wide spectrum of pollutants, Lead (Pb) and Tin (Sn) were extensively removed, along with trace amounts of Copper (Cu) and Zinc (Zn). The presence of Silicon (Si), Magnesium (Mg) and Carbon (C) was also detected in the adsorbent.
As compared to other available treatment methods, this method proposes a reduced treatment time. “Our technique takes about 30 minutes, whereas it can be several hours or even days in other methods. We have also used a single step process instead of conventional multi-stage approaches”, says Prof Khanna.
These researchers are now working towards optimizing this process and developing a variety of adsorbents to reduce toxic elements in the outgoing gases produced during high temperature treatment of e-waste. “Our main focus is on minimising the environmental damage, especially to the areas surrounding the waste processing facilities. Release of dust particles, toxic fumes, metal particulates, dumping of residual non-valuable materials are some of the key issues we sought to address. Overall costs of e-waste processing are expected to be quite low using our technique. I would expect these to be much lower than current e-waste recycling approaches”, adds Prof Khanna, reiterating the benefits of this approach.