Our body heals an accidental injury, a knife cut for example, within a few minutes. Have you ever wondered how that happens? Immune cells migrate to the site of injury and subsequently heal the injury. The active migration of blood cells towards the cut site is an example of directed migration. But how do cells sense and respond to directional cues like an injury?
Two collaborative studies that spanned over three consecutive summers at the Marine Biological Laboratory (MBL), Woods Hole, USA comprising researchers from NCBS Bangalore, National Institutes of Health and Harvard Medical School, USA has provided new insights into the process of directed cell migration [1] [2].
How do cells migrate in the first place?
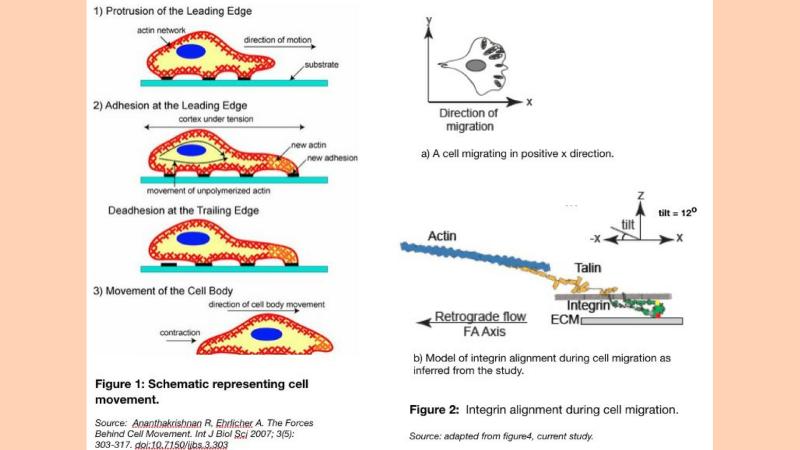
Cell migration is a key process in the development and maintenance of multicellular organisms. Wound healing, immune responses and tissue formation during embryonic development are some of the processes that require the directed movement of cells to specific locations. Proteins present on the cell’s surface, called integrins, sense external cues like injury, and transmit the information to the cell’s internal machinery. The actin filaments – long fibres of the cytoskeleton that span the inside of a cell and are made up of a type of globular protein, form long protein chains and along with motor proteins generate forces that ultimately result in the propulsion of the cell towards the external cue.
The integrins reside within aggregations of multiple proteins called focal adhesions- structures that enable the cell to bind to its extracellular substrate. Contraction at the rear end, or the part of the cell away from the cue, and the release of focal adhesion complexes help in the migration of cells towards the external signal (Figure 1).
Thus, cell migration involves extensive communication between the cell’s surface and its interior. An important link in this communication are the integrin proteins. The integrins transduce the force generated inside the cell to the substrate outside the cell and thus facilitate cell movement.
“Integrins are proteins on the cell membrane which sense environmental cues and regulate cell function”, said Joseph Matthew K, one of the lead authors of the study. “Our study provides the first evidence that integrins can also sense directional cellular forces and align themselves in response to those forces.”
How do cells determine the direction of migration?
Focal adhesions have been shown to respond to directional physical cues. However, how is this infomration tranduced to the cell at a molecular level? Do integrins provide directional information and could they function as the elusive ‘direction encoder’ that operate within focal adhesions? Indeed. Integrins own activation status is directly coupled with structural changes of the molecule.
Previously published studies of integrins have identified an “activated state” – a structural conformation where integrin proteins are fully extended and can easily bind to the substrate. However, how this active state is induced is still not understood. Though several competing theoretical models of integrin activation exist, the current study provides credible evidence of the ‘cytoskeletal force model’, which states that a force exerted by the actin cytoskeleton is required for extending and activating the integrin molecule. Since actin flow is directional, the coupling of this flow to the activation status of the integrin molecule could encode directional information in the cell.
For the study, the researchers used highly motile fibroblast cells from mouse as a model system. To study the organization of integrin proteins on fibroblasts’ surface during migration, the team fused a constraint fluorescent protein to integrin. This enabled visualizing the orientation of integrin molecule within focal adhesion complexes. One of these methods based on fluorescence emission polarization was developed in the Mayor laboratory in Bangalore and the other - the PolScope was developed by Tomomi Tani and Rudolf Oldenburg at the MBL.
The authors found that integrin molecules within a focal adhesion protein complex are co-aligned. Furthermore, the alignment of integrin molecules on the cell surface was strongly correlated with the alignment of actin filaments inside the. This ordering was dependent on the linkage of the integrin to the actin cytoskeleton via linker proteins called talins.
Previous studies have shown that when the cell extends its cell membrane to move forward, actin filaments inside the cell move in the opposite direction (retrograde flow). This retrograde flow generates the necessary forces that facilitate cell movement. To test if actin retrograde flow is required for proper integrin alignment, the researchers blocked the flow using inhibitors. They found that in regions where the retrograde actin flow was completely blocked, the integrins alignment was altered. In regions inside the cell, where actin flow remained intact, integrins were aligned as in the normal migrating cells.
The polarization microscopy images gave a 2D projection of integrin alignment on the cell membrane. The researchers next used Rosetta modelling, which is a sophisticated program for computation and modelling of protein structure, to infer the 3D orientation of integrin proteins inside focal adhesions. They find that not only was the integrin aligned in the direction of actin flow but it also adopts a tilted conformation (10 degrees) with respect to the substrate surface (Figure 2). By this exercise, they ellucidated the kind of a conformational change that is required for the protein to achieve the activated state, which was not known before.
The same group of scientists also looked at the orientation of integrins in fast migrating immune cells known as T-cells and observe a similar correlation between the alignment of integrins and the direction of actin flow at the leading edge of the migrating cells.
Taken together, these studies show that directional actin retrograde flow inside the cell is conveyed to the integrin proteins on the cell membrane, which result in the activation and co-alignment of integrins within the focal adhesions and facilitate directed cell movement.
“We will continue to investigate how directional cellular forces affect focal-adhesion function. A prediction from this study would be that only those ligands that are aligned or those tracks that line up with the direction of the forces are likely to be strengthened and also the ordering of molecules in focal adhesions would restrict interactions amongst proteins within the focal adhesion complex, signs off Prof Mayor, one of the senior authors on the paper.