Research Matters is happy to bring you this article as part of the series on Mathematical Modeling and Data Analysis by the Mathematical Modeling team of Indian Scientists' Response to Covid-19 (ISRC). The first part of this series is on Explaining Models of Epidemic Spreading.
Why do we need Mathematical Models for CoVID-19?
In an event like an epidemic, policymakers are keen to know how the disease will spread. For instance, they might be keen to know how many people are likely to be infected in future. This will help them make decisions and allocate resources towards disease control, like the number of ICUs or ventilators required in a region. These and several other predictions concerning the spread of disease are accomplished using mathematical models of epidemiology.
Mathematical models help us make our mental models more quantitative. Models are not reality; however, studies across the natural and physical sciences have shown the importance of models in understanding nature. Say, we need to send a spacecraft to the Moon. To find out how much velocity a spacecraft needs for it to escape the Earth’s gravity, we would not design hundreds of spacecraft and launch them at different speeds to see which one reaches the Moon, right? Instead, we rely on mathematical equations that clearly predict the velocity and all other features that a spacecraft should have, to reach the Moon.
All said, models are not always accurate representations and they do come with limitations. To extend the analogy of the spacecraft designed to reach the Moon, if it were to reach Jupiter, our model would need some tweaks to get it there. It is important to understand the assumptions behind a model and its scope before using it to make predictions and policies.
Models are used to predict the future of the system under study. In the case of epidemics too, we need mathematical modelling to understand how the disease is most likely to spread, and where it is more likely to spread. It can be viewed as a shortcut, instead of implementing many guesses about how to deal with the spread of a disease, we can see what implementing each of these guesses would mean, using some nifty equations, and take more well-informed decisions. Even as you read this, mathematical modelling has been at the heart of several policy decisions worldwide regarding the response to CoVID-19.
Some Aspects of Mathematical Models
So, the question is, how are models developed and used? Typically, models are constructed based on some reasonable hypotheses. For instance, to construct a model for epidemics, one needs to make some assumptions about the mode of disease spread. Measles or Covid19 spread when infected individuals come in contact with healthy ones. Malaria on the other hand needs both mosquitoes and infected humans. A model for malaria would need to take into account the mosquito population as well as that of people and would differ greatly from a model for measles. To give another example of a hypothesis that goes into model formulation, one could postulate age-dependent infectivity, i.e., that the likelihood of being infected on contact, is dependent on the age of the person, with older people more likely to contract the infection. Similarly, one could postulate something about recovery (“younger people recover faster”) or reinfection (“a person who recovers is immune from reinfection for a period of two years”). All these hypotheses can be explicitly incorporated into models. These hypotheses are based on the biology of the disease, i.e., on what we know about the pathogen and the human body. Several different models for the same disease are possible, each differing in the finer details it incorporates among its hypotheses. Needless to say, models are only as good as the assumptions they are based on.
All models have parameters – numbers which can be tuned to suit the particular context to which the model is being applied. For example, consider control measures such as physical distancing. Under normal circumstances, people tend to be physically near each other, leading to a greater likelihood of being infected on proximal contact. When physical distancing is enforced, this likelihood decreases. The likelihood of infection on contact is thus a parameter; when chosen to be small, it captures the situation where physical distancing is being enforced and when chosen large, it captures the business-as-usual scenario. Similarly, age demographics vary by country, state or region. If age-dependent infectivity is part of our model hypothesis, we would need parameters which keep track of the proportion of population in each age-group. These parameters would have markedly different values for India and the US, for example; The Indian population is predominantly young, while that of the US is more evenly spread out across ages. Recovery rates or immunity periods are also parameters, which can be tuned differently to model different diseases. Thus, choosing parameters appropriately allows one to apply the same underlying model to different diseases, different countries or regions, under different control measure scenarios etc.
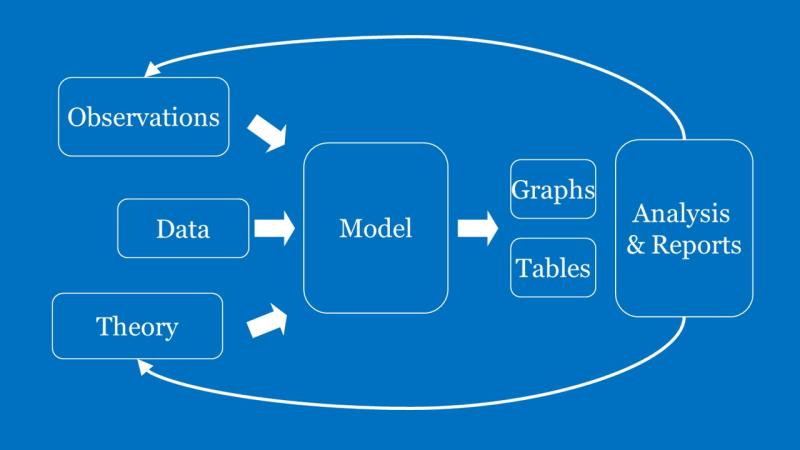
Once the broad hypotheses and parameters are chosen, the model is written down in terms of mathematical equations. These are typically differential equations, which can then be solved on a computer to obtain the quantities of interest (for eg, the number of infected people) at different points of time.
At the outset, we cannot be sure that we’ve made good choices of parameters. But we correct this slowly by comparing model predictions with actual data (as it becomes available) and tuning our parameters so that there is a close match between these. For instance, if we want to use a new model to predict the number of CoVID-19 infections in Chennai in June 2020, we first validate it using the data on infections until now. In other words, we fit our parameters such that our model is able to explain the daily number of infections until today (April 16, 2020). Once the model is validated, it can be used to predict future behaviour and suggest new experiments to study the population. As days go on and new data becomes available, it is possible to test the model predictions. In some cases, the model is improved/refined as more data becomes available and the cycle continues.
Coming soon — SIR and SEIR Models of Infectious Diseases.
Editor's Note: This was originally published by Indian Scientists Response to Covid-19 (ISRC). Republished here with permission.






