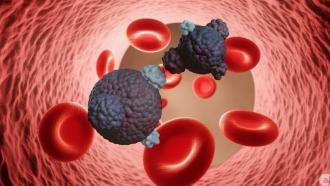
Our brain, the seat of our personalities, memories and emotions, is arguably the most critical organ in our body. If a cancerous tumour grew in our brain, it might impair its functions, and the results would be catastrophic. While chemotherapeutic drugs are used to combat cancer, these drugs also damage healthy cells, restricting their growth and reproduction. In an attempt to make chemotherapy safer, researchers at the Indian Institute of Technology Kanpur have proposed a new model for the transport of anti-cancer drugs to human brain tumours using liposomes—a small sac-like structure that transports nutrients in our cells.
Liposomes, like our cell membranes, are made of molecules called phospholipids. In a cell membrane, these molecules are arranged into two layers. However, in a liposome, the two layers meet to form a hollow sphere filled with water. In this study, the researchers propose to replace the water in the empty area with anti-cancer drug molecules—a technique they claim improves transport of the drug molecules—to create a nanosized drug delivery system.
Besides, the lipid layer in the liposomes acts as a protective coat and prevents the anti-cancer drug from affecting healthy cells. Since these liposomes are in the bloodstream, they circulate for longer and collect at the tumour site, helping more of the drug reach their target cells. This approach causes less damage to the healthy cells of the body.
“Liposome-encapsulated drugs are widely used to cure cancer because of their prolonged circulation time in blood plasma and better efficacy as compared to free chemotherapeutic drugs”, says Dr. Ajay Bhandari, a research scholar at IIT Kanpur and an author of this study, published in Journal of Biomechanical Engineering. “DOXIL, a type of liposome encapsulated drug that has been widely used to treat different types of human tumours, has been beneficial towards improving chemotherapeutic drugs, making them more efficient and safer to use”, he adds.
There is another aspect of tumours that makes it hard to use anti-cancer drugs in treatment without damaging the surrounding healthy cells. Tumorous cancer cells vary in their organisation, function and structure, making it difficult for drugs to target them specifically. Also, the veins and arteries that supply blood to a tumour, twist and coil, making it more difficult to administer the anti-cancer drug into a tumour. Since the endothelial cells present in the tumour blood vessels are widely spaced out, drugs tend to leak out into the interstitial spaces between the cells, reducing the efficacy of the drugs.
“Liposome-encapsulated drugs can accumulate at the tumour site better than a free chemotherapeutic drug because they can circulate in human blood plasma for a longer amount of time. The drug DOXIL, used for simulation in our current study, does not get cleared out from the human body easily. Also, since liposome-based anti-cancer drugs have a higher concentration in the blood plasma, they tend to have a higher concentration in the interstitial space, thus killing more tumour cells”, says Dr. Bhandari, talking about the proposed drug-delivery technique.
The researchers analysed the structure of blood vessels of the cancerous brain tissue using dynamic contrast enhanced magnetic resonance imaging (DCE-MRI) by taking multiple snapshots of the tumor vasculature over a period of time and comparing them. Using this data, they predicted the transport, accumulation and efficacy of the chemotherapeutic drug in a tumour with computational fluid dynamics—a branch of fluid mechanics that uses numerical analysis and data structures to solve and analyse problems that involve fluid flows. They also predicted the distribution of the drug in a tumour. “With the help of this model, we can predict the preferential deposition of chemotherapeutic drugs inside a tumour, thus predicting the healing of a tumour for a specific patient, provided we have the initial medical images of that patient”, says Dr. Bhandari.
The results of the study showed that a high amount of the anti-cancer drug Doxorubicin, which was not encapsulated by a liposome, accumulated in the interstitial spaces between tumour cells. This accumulation could potentially cause adverse side-effects like swelling, reddening and peeling of skin from the hands and feet. In contrast, the liposome-encapsulated drug remained inside a tumour for a longer time. They also found that the high concentration of liposome-coated drug aggregated at the tumour site and increased the therapeutic efficiency of the liposomal anticancer drug.
Medical imaging of the tumours helps predict the deposition of the anti-cancer drug in a tumour for a patient and look for chemotherapeutic drug options that could better suit him/her. This ability also allows doctors to develop a specific chemotherapeutic approach for each patient, based on the structure of their brain tumour and the blood vessels surrounding it.
The researchers hope that medical imaging, combined with numerical models, can help cancer patients better by reducing the adverse side-effects seen with many drugs. “To this end, numerical modelling is an excellent non-invasive tool to understand the complexity that is involved in the transport of anti-cancer drug”, says Dr. Bhandari, talking about the importance of the research.