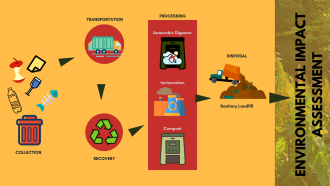
After graphene, carbon nanocones are now an exciting form of carbon for material scientists. As the name suggests, they are conical structures made up of carbon, where graphene sheets are folded like a party cap with a height and diameter of a few nanometers (1 nanometer = 1/1,000,000,000th of a metre). With unique properties due to their conical shape, they have a wide range of applications, including being used as a tip of the probe of a high precision microscope used to record activity at an atomic level. However, studying the properties of an individual nanocone is a challenge since they tend to accumulate in stacks. In a new study, researchers from the Indian Institute of Technology, Kanpur have simulated the separation of a single carbon nanocone from a nanocone stack.
In this study, published in the journal Carbon, the researchers have studied the various mechanical forces acting on the nanocone during this process and have predicted the types of deformations that could occur in the structure of the nanocone.
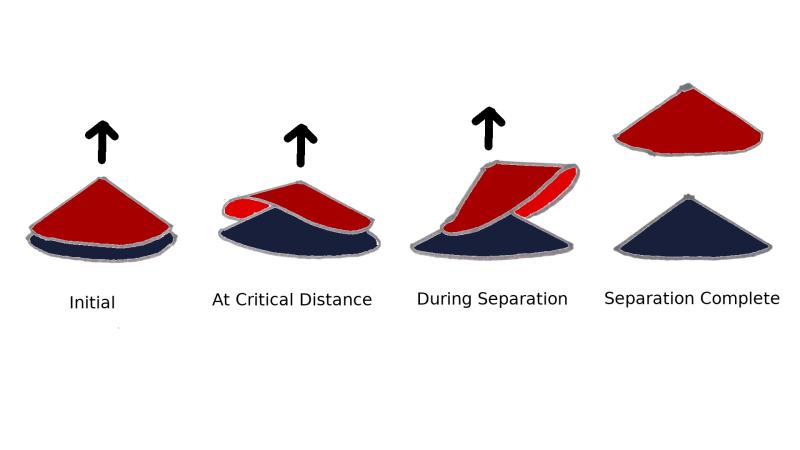
For the simulations, the researchers considered the cones stacked in a way where their tips are along the same vertical line. They then fixed the base of the bottom-most cone on a substrate, and gradually pulled off the top-most cone in tiny steps along the vertical line. At each stage, the researchers considered all possible forces acting on the system, calculated various components of energy in the system and studied the shape of the top and the bottom cones.
The researchers observed that the total energy of the system gradually increased during the process of pulling, but at a distance of 0.43 nanometers between the top-most cone and the one below it, this energy dropped suddenly. The researchers also observed that the forces acting on the cone fluctuated. At this stage, the wall of the upper cone bent due to compressive forces and deformed outwards, and its rim almost reached the height of its apex. The rest of the rim stayed attached to the bottom cone due to van der Waal’s forces—attractive forces between molecules that become significant when they are very close to each other.
The deformation, the researchers say, was due to the competing forces—the pull that tends to stretch the rim wall and the attractive molecular force that tends to keep the molecules in the wall close to each other, and to the wall of the cone below it. When the researchers pulled the top-most cone further away, it gradually started recovering its shape and was back to its original form after separation.
The deformation, the study notes, occurs at any temperature. The researchers simulated the process of separation at two different temperatures—first at a value close to ‘absolute zero’—when the atoms vibrate the least, and the second, at room temperature of about 27o C. They also repeated the simulations for the action of pushing the cone back on the stack and observed that the cones flipped inside out.
The findings of the study can help experimentalists engaged in the research of carbon nanocones. It demonstrates that during the process of separating nanocones, they could deform, and the device used to pull the cone could experience a jerk at the critical distance. This knowledge can help material scientists to slow down the speed of pulling out a nanocone or take appropriate measures to avoid any likely damage.