![Image for representation purposes only [Image Credits: Tom Ellenberger, Washington University School of Medicine in St. Louis. / Public domain] The vital role of Plasmodium’s molecular scissors in its cellular function](/sites/researchmatters/files/styles/large_800w_scale/public/molecule_12june.jpg?itok=vW_of5D_)
The nucleus of our cells contains the blueprint of life—the DNA—tightly wrapped around by a layer of proteins called histones. These proteins, arranged like beads, together with the DNA, form the chromatins. This arrangement suppresses the synthesis of new proteins when the body does not need them. However, when we do need them, a particular class of 'worker proteins' must bind to the DNA by changing the chromatin structure and unwinding the DNA from the histone beads. The addition of an acetyl group to the histones helps this process, which frees up the DNA and leads to the production of the needed proteins.
In a recent study, researchers at the Jawaharlal Nehru University, New Delhi, have found a new mechanism of controlling protein synthesis in the malaria-causing parasite, Plasmodium. They find that specific enzymes, called histone acetyltransferases (HATs), undergo a unique protein-processing pathway that might influence the overall gene expression in these parasites. Their study was published in the Journal of Cell Science and funded by the Department of Biotechnology (DBT), Science and Engineering Research Board (SERB) and University Grants Commission (UGC).
In Plasmodium, the histone acetyltransferases add acetyl groups (CH3CO) to the histone proteins and help the parasite in moving forward from one stage of its life cycle to another, in different hosts. When Plasmodium shifts from the belly of a female Anopheles mosquito into our liver cells, and then to our red blood cells, the environment at each of these hosts is different. Among the several other essential factors, these enzymes help the parasite to fight our immune system and thrive in our cells.
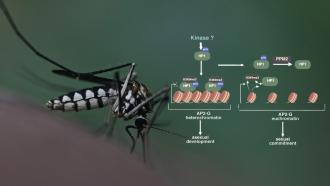
The researchers of the current study focused on the protein HAT-GCN5 in Plasmodium. This protein is needed to enter human cells and cause severe malaria. Although the role of GCN5 in causing the infection was known, ways in which it was self-regulated and governed was mostly unknown. The researchers began by isolating GCN5 from the pool of Plasmodium proteins. They found that this protein existed in multiple sizes, and truncated versions of GCN5 hinted that it could be cut into smaller fragments. But which molecular scissor was behind this? That's the question the study finally answered.
Akin to how a hairdresser, a tailor and a doctor all use scissors—although of different shapes and kinds—some enzymes in our body also can cut molecules. These enzymes, called proteases, have specific functions based on their shape, size and location in the cell. For example, a cysteine protease cuts just after the cysteine amino acid in its target protein. The researchers of the current study did not know which specific molecular scissor was being used in Plasmodium to chop GCN5. Hence, they sequentially blocked one known class of protease at a time, until the protein remained uncut in the cell. They figured out that blocking cysteine protease/s did not result in the trimming of GCN5.
The researchers then used fluorescence microscopy to identify the exact location in the cell where this trimming was taking place. They found a pile-up of uncut GCN5 molecules in the lysosomes. This cell organelle stores proteases, which are necessary for the degradation of haemoglobin. The study also found that GCN5 proteins in Plasmodium were synthesized on the endoplasmic reticulum—a network of membranes in the cell. They then enter the Golgi, responsible for transport, and move to lysosomes, where they are cut. Eventually, they reach the nucleus and add the acetyl group to the histones, setting the DNA free for protein synthesis.
"Our work signifies that these cysteine proteases are possibly capable of processing other proteins. They also play a vital role in the parasite other than haemoglobin digestion," says Mr Krishanu Bhowmnick, the lead author.
The study found that the unprocessed protein is trafficked to the food vacuole via the ER-Golgi secretory pathway.
"This kind of constitutive trafficking of a chromatin-modifying enzyme through the ER-Golgi route has not been reported in any other organisms", he adds.
The findings show how vital the trafficking pathway of the GCN5 proteins and its molecular scissor is. If cysteine protease is blocked, the protein does not reach the nucleus, failing to add the acetyl group needed for protein production. These proteins are necessary for Plasmodium to multiply and survive.
"The study unravels a unique pathway for parasite biology that may serve as a potential target for therapy," signs off Prof Suman Kumar Dhar, the corresponding author of the study.
This article has been run past the researchers, whose work is covered, to ensure accuracy.