A small depression in the football field, invisible to the eye due to levelled grass, may have given you a foot twist twice. The third time, however, you evade it like a charm because your brain has been constructing a map of the field and alerts you about the danger. Tiny cells called neurons, with their long tails and fibrous fingers, link together to form pathways within the brain that help you learn about your surroundings. However, it is difficult to pick out a single pathway to understand its function as the brain is chock-a-block with interconnected neurons.
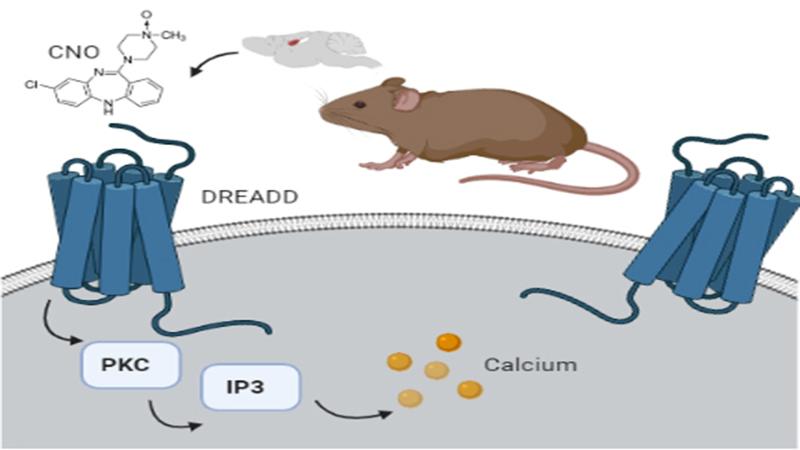
Chemogenetics is a technique that uses chemicals to understand how neurons are activated. It manipulates genes to place the neurons under the control of special receptors called DREADDS. Short for Designer Receptors Activated by Designer Drugs, these receptors only bind to an inert chemical called Clozapine-N-Oxide (CNO). In a recent study, researchers from the Tata Institute of Fundamental Research (TIFR), Mumbai, and Jawaharlal Nehru Center for Advanced Scientific Research (JNCASR), Bengaluru, point out specific flaws in this technique. The study was published in the journal eNeuro and funded by the Department of Science and Technology and Scientific and Engineering Research Board.
“We started these series of experiments because we observed differential behavioural effects in mice administered different doses of Clozapine-N-Oxide,” says Dr. Sthitaprajnya Pati, the lead author of the paper. He is a postdoctoral fellow at TIFR.
The researchers used a designer receptor named hM3Dq for their study. When this receptor interacts with CNO, it increases calcium ions within the cell, which then induce the release of chemical messengers called neurotransmitters. An activated neuron releases its neurotransmitter at the synapse—a point of contact with another neuron. Neural circuits or pathways are formed by multiple neurons connected via synapses. Activation of one neuron generates a ripple that is transmitted across different neurons communicating with each other within a circuit.
But how should one know what dose of CNO must be used to activate a neural circuit?
“Different doses of CNO can have very different pharmacological effects,” explains Dr Pati. “They may include the use of differential downstream signalling pathways and cross-talk with other receptors. It is also possible that the metabolism and effect of CNO on the body can vary across strains, species, sex and age”, he adds.
The researchers turned to the hippocampus in the brain to understand the effects of different doses of CNO. Hippocampus, a creatively-named seahorse-shaped structure, plays a vital role in memory formation. The researchers used hippocampal slices from genetically modified mice, where the designer receptors were present on the primary excitatory neurons of the Schaffer Collateral pathway. This pathway is essential for memory formation as it connects different parts of the hippocampus. Since the primary excitatory neurons carry most of the information along this pathway, controlling them by chemogenetics may reveal a lot about their functioning.
The researchers measured the changes in neuronal properties after 1µM and 20µM dose of Clozapine-N-Oxide. They observed that different doses of CNO had complex and varied effects on the neurons. A low dose of CNO increased the chances of neuronal activation and a high dose led to a reduction in neuronal excitability.
Neurons have a specific voltage maintained by the difference in the amount of positively and negatively charged ions inside and outside the cell. Its membrane houses particular channels for the charged ions to move in and out of the cell. A change in voltage can open the ion channels leading to an inflow of positively charged ions and activating the neuron. The channels remain open until the ionic balance returns to normal and the voltage stabilizes.
The researchers discovered that a dose of 20µM CNO caused a steady reduction in the incoming positive currents mediated by the ion channels, whereas a 1µM dose caused a weaker effect. Intriguingly, the effect of the high dose lasted at least an hour after CNO was completely removed from the hippocampal slices.
By controlling neuronal activation via designer receptors within the hippocampus, one can track the electrical and chemical changes involved in long-term potentiation and memory formation. Long-term potentiation is a process where connections between neurons are strengthened, which help in generating memory. The researchers observed that high doses of CNO prevented long-term potentiation, while low doses facilitated it. Hence, different doses of CNO can have almost opposing effects on a neuron.
As the application of chemogenetics extends beyond rodents into nonhuman primates, these results urge us to interpret the data under a new light.
“Our data elucidates that DREADD activation can not be used as a simple proxy for neuronal excitation,” says Dr Pati. “Rather, it initiates a plethora of downstream changes that vary both in space and time. Our suggestion is to be careful while selecting the CNO dose and time after which behaviour is carried out”, he cautions.
The findings of the study would be useful for researchers working to develop the next generation chemogenetic tools as they can work to remedy these shortcomings and variability of the engineered receptors.
This article has been run past the researchers, whose work is covered, to ensure accuracy.






